COVID-19 RAPID TESTS
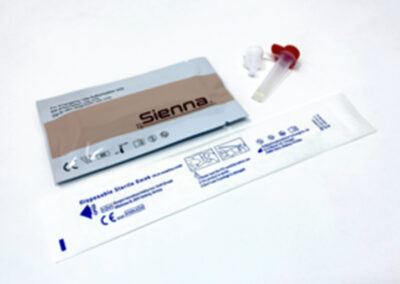
Sienna™ COVID-19 Rapid Antigen Test(Box of 25)
COVID-19 Rapid Test
Sienna™ COVID-19 Rapid Antigen Test
The Sienna Covid-19 rapid antigen test is authorized for professional use by the FDA (EUA) in point-of-care settings by organizations with CLIA certification.
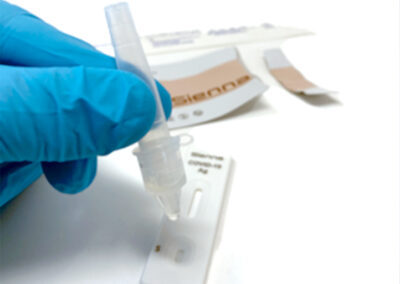
With visual results in 10 minutes and nasopharyngeal collection, Sienna is fast and accurate.
General Details
-
-
-
Test type: Antigen
-
Run time: 10-20 minutes
-
Collection type: Nasopharyngeal swab
-
Authorization: FDA EUA for professional use. CLIA required
-
Intended Use: Lateral flow immunoassay intended for the qualitative detection of SARS-CoV-2 nucleocapsid protein.
-
Manufacturer: Salofa Oy (Finland)
-
-
Box components
-
-
-
Nasopharyngeal swabs (25)
-
Test cassettes (25)
-
Extraction reagent capsules (25)
-
Workstation for mass testing (1)
-
Instructions for use (1)
-
-
iHealth® Home Test
Manuals + Instructions
Intended Use
Qualitative detection of nucleocapsid protein antigen from SARS-CoV-2 in direct anterior nasal swabs from individuals suspected of COVID-19 by their healthcare provider within the first five (5) days of symptom onset. Emergency use of this test is limited to authorized laboratories.
In the USA, the SPERA COVID-19 Ag Test has not been FDA cleared or approved; but has been authorized by FDA under an EUA for use by authorized laboratories; use by laboratories certified under the Clinical Laboratory Improvement Amendments of 1988 (CLIA), 42 U.S.C §263a, that meet the requirements to perform moderate, high, or waived complexity tests. The SPERA™ COVID-19 Ag Test is authorized for use at the Point of Care (POC), i.e., in patient care settings operating under a CLIA Certificate of Waiver, Certificate of Compliance, or Certificate of Accreditation.
Clinical Details
-
SARS-CoV-2: 93.8% Sensitivity, 99.2% Specificity
-
Specificity: No cross-reaction with species tested
-
Interference: No interfering factors tested
-
CPT Codes: Covid-19 87811QW
Storage and handling
Test kits should be stored at 2-30°C (35-86°F). Reagents and materials are stable until the expiration date printed on the outer packaging. Do not use beyond the expiration date. The test device must remain in the sealed pouch until use. Do not freeze any contents of the kit.
CONTACT
Direct Connect
Phone
(310) 957-9891